Raw manuka honey contains methylglyoxal and hydrogen peroxide with demonstrated activity against Streptococcus mutans and Porphyromonas gingivalis — the key bacteria behind tooth decay and gum disease. Studies suggest it may reduce plaque formation and support gum health when used as an adjunct to regular dental care. It does not replace brushing, flossing, or professional dental check-ups.
Your mouth is home to more than 700 species of bacteria. Most cause no trouble. One does: Streptococcus mutans, a sticky, acid-producing organism that builds protective biofilm on tooth enamel and is the primary driver of dental decay. Standard antimicrobial mouthwashes disrupt it. So does methylglyoxal: the compound that makes high-grade mānuka honey biologically different from every other honey on the market.
This article covers what the peer-reviewed research actually shows about mānuka honey and oral health, where the evidence is solid, where it is preliminary, and where it is still in vitro only. The science is genuinely interesting. It also has limits, and I want to be honest about both.
The bacteria in your mouth work harder than your toothbrush
Within hours of brushing, Streptococcus mutans begins rebuilding dental plaque on tooth surfaces. It does not float passively in your saliva. Instead, it secretes extracellular polysaccharides that form a structured matrix: a biofilm that physically shields bacteria from antimicrobial agents and concentrates the acid they produce against enamel. This is why plaque is sticky and why rinsing with mouthwash alone cannot remove it.
The acid in question is lactic acid, produced when S. mutans ferments dietary sugars. At the enamel surface, lactic acid drives demineralisation: it dissolves calcium and phosphate ions from the crystalline structure of the tooth. If this process outpaces remineralisation, the result is a cavity. Fluoride helps restore the balance; most antibacterial interventions address the bacterial load driving the process.
Gum disease involves a different but related community. Porphyromonas gingivalis, Fusobacterium nucleatum, and Prevotella intermedia are anaerobic gram-negative bacteria that colonise the gingival sulcus: the narrow groove between tooth and gum. These organisms trigger the chronic immune response that characterises periodontitis, the inflammation that, if untreated, progressively destroys the bone and connective tissue holding teeth in place.
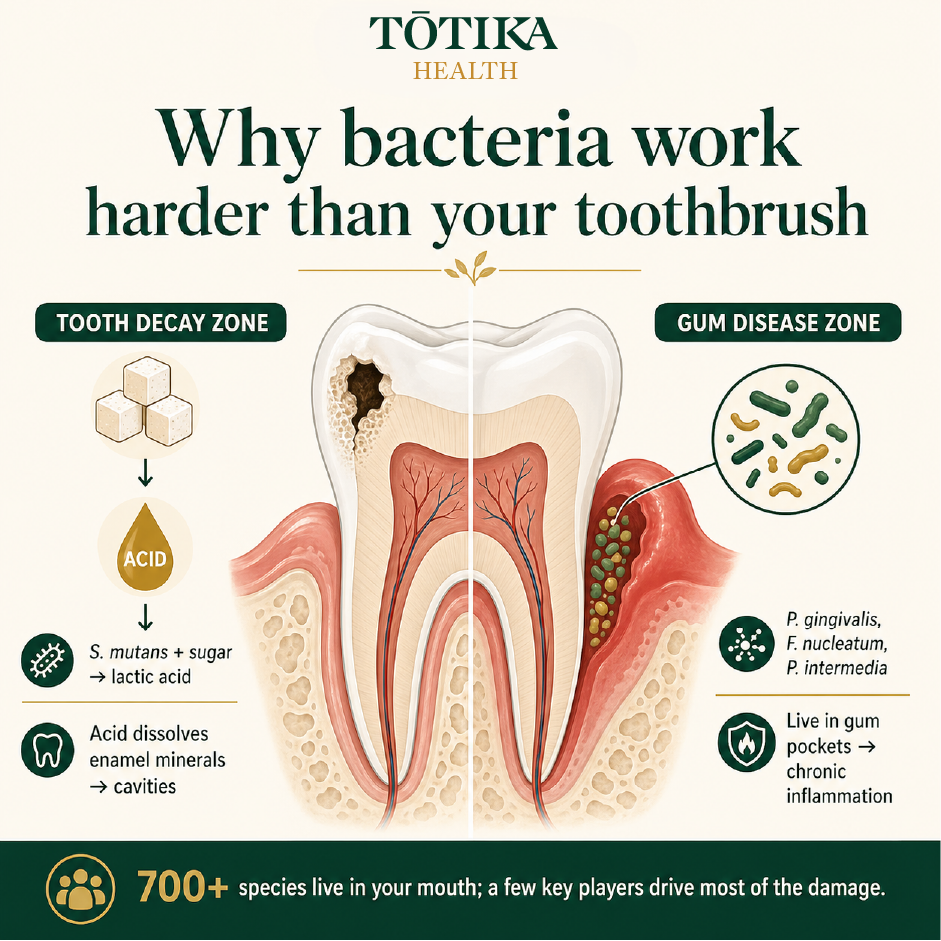
700+ species live in your mouth. A handful drive most of the damage: one zone eroding enamel, another destroying the tissue holding your teeth in place. Source: Tōtika Health.
What MGO actually does to oral bacteria
Methylglyoxal (MGO) forms naturally in mānuka honey from dihydroxyacetone (DHA), a compound present at high concentrations in the nectar of Leptospermum scoparium. As honey ripens in the comb, DHA converts progressively to MGO. This is why raw mānuka honey that has been allowed to fully mature contains significantly higher MGO than honey harvested early in the season: the transformation takes time, and prematurely extracted honey leaves MGO potential unrealised. "Raw" here means unprocessed and fully matured in the comb. That is chemistry, not marketing.

The antibacterial difference starts in the nectar. DHA converts to MGO as the honey matures in the comb. Harvest too early and the potential is lost. Source: Tōtika Health.
MGO's antibacterial mechanism is distinct from the hydrogen peroxide activity found in most conventional honeys. Hydrogen peroxide is unstable in the warm, enzyme-rich environment of the mouth, where catalase enzymes from saliva and bacteria rapidly break it down. MGO is stable under the same conditions. It disrupts bacterial cell membrane integrity, inhibits key enzyme activity, and at sufficient concentrations causes irreversible DNA damage in susceptible organisms.
Methylglyoxal (2-oxopropanal) — the active molecule in mānuka honey
Methylglyoxal has exactly 3 carbons. The ketone and aldehyde groups sit directly adjacent (shown inside the dashed gold bracket), which is chemically unusual and the source of MGO's high antibacterial reactivity. Most compounds have only one reactive site.

Two adjacent reactive sites make MGO structurally unusual and highly effective against bacterial proteins. Regular honey has one. The difference is molecular, not cosmetic. Mechanism: Safii, Tompkins and Duncan, University of Otago, 2017 [2].
In vitro research from the Sir John Walsh Research Institute at the Faculty of Dentistry, University of Otago, tested medical-grade mānuka honey against twelve species of oral bacteria, including S. mutans, P. gingivalis, F. nucleatum, and P. intermedia [2]. The honey demonstrated antibacterial activity against all twelve species tested. This is laboratory evidence, not a clinical trial, but it confirmed that mānuka honey reaches a biologically relevant range of oral pathogens, not just the organisms most commonly studied in isolation.
A randomised, double-blind clinical study published in 2023 compared a mānuka honey mouthwash against 0.2% chlorhexidine and distilled water in 45 participants over 21 days [4]. The mānuka honey group showed statistically significant reductions in both the plaque index and salivary S. mutans bacterial load compared to baseline. Chlorhexidine was the more effective agent overall, as you would expect. But the mānuka honey group achieved meaningful reductions without reported side effects, which is clinically relevant when considering something used repeatedly over months.
What the gum disease research shows
The most clinically meaningful published trial to date on mānuka honey and periodontal disease is a 12-month, split-mouth pilot study from the University Hospital Zagreb, published in Materials in 2023 [3]. Split-mouth design means each participant served as their own control: one half of the mouth received standard non-surgical periodontal therapy (NSPT) alone; the other half received NSPT plus mānuka honey applied directly into the gingival pockets by the clinician.
At 3, 6, and 12-month follow-up points, the mānuka honey quadrants showed statistically significant improvements in periodontal pocket depth (PPD) compared to NSPT alone. Clinical attachment level, a measure of how much supporting tissue has been lost, also improved significantly at 3 and 6 months. The authors appropriately note that this is a pilot study of 15 participants and that the findings require confirmation in larger randomised controlled trials before firm clinical guidance can be issued.
What is notable from a research design standpoint is the 12-month follow-up. Many adjunctive periodontal interventions show promising short-term improvements that do not hold at one year. The persistence of pocket depth improvement here is worth tracking as larger studies are conducted.
The University of Otago study you should know about
The foundational clinical research connecting mānuka honey to dental plaque and gingivitis control was published in 2004 by English, Pack, and Molan at the University of Otago School of Dentistry [1]. Thirty participants were randomised to either chew a mānuka honey preparation (a chewable “honey leather” made with UMF 15 honey) or sugarless chewing gum three times daily for 10 minutes, over a 21-day period.
The mānuka honey group showed statistically highly significant reductions in mean plaque scores (from 0.99 to 0.65, p=0.001) and in the percentage of bleeding sites (from 48% to 17%, p=0.001). The control group showed no significant changes. No significant differences in caries activity were observed. The small sample size and short duration limit generalisation, but as a randomised controlled trial, this study established that high-activity mānuka honey held in contact with teeth and gums can produce measurable improvements in plaque and gingival health.
The Otago faculty connection to this area of research is ongoing. The 2017 in vitro study by Safii, Tompkins, and Duncan at the same institution built directly on this foundation, testing antibacterial activity across twelve oral pathogen species [2]. A 2025 in vitro study from the University of Bern also reported significant biofilm disruption from mānuka honey and propolis toothpaste formulations, with the propolis-mānuka combination producing the strongest reduction in colony-forming units within established biofilms [5].

Three studies, three formats, one consistent direction. Chlorhexidine remains the stronger antimicrobial. Mānuka honey's case rests on adjunct benefit and tolerability. Sources: [1] [3] [4].
Bad breath and what the evidence actually supports
Halitosis caused by oral bacteria is driven primarily by volatile sulphur compounds (VSCs), produced by anaerobic gram-negative species colonising the posterior tongue, the gingival sulcus, and periodontal pockets. The same organisms implicated in periodontitis, specifically P. gingivalis and F. nucleatum, are significant VSC producers.
The 2023 Al-Kubaisi clinical study documented broader reductions in oral bacterial load in the mānuka honey mouthwash group [4]. Halitosis was not directly measured as an outcome, so improved breath remains an indirect inference rather than a demonstrated trial result. There is no published randomised controlled trial specifically on mānuka honey and halitosis. What is reasonable to say is that improving gingival health and reducing the bacterial load of VSC-producing organisms may contribute to improved breath as a secondary effect. That distinction matters.
How the options compare
| Factor | Regular honey | Mānuka MGO 514+ | Chlorhexidine 0.2% |
|---|---|---|---|
| Primary antibacterial mechanism | H₂O₂ (unstable in mouth) | MGO (stable, non-peroxide) | Cationic membrane disruption |
| Active against S. mutans | Moderate (in vitro only) | Yes (clinical and in vitro) | Yes (extensive evidence) |
| Active against periodontal pathogens | Limited | Yes (University of Otago, in vitro) | Yes |
| Disrupts established biofilm | Low | Moderate to strong (in vitro) | Strong |
| Long-term use concerns | Sugar content in quantity | None reported in studies | Tooth staining, taste alteration, microbiome disruption |
| NZ clinical research | None | Yes (University of Otago, 2004 and 2017) | Extensive global evidence base |
How to incorporate mānuka honey into your oral health routine
The research does not yet support a single standardised home protocol. What the published studies share is a common principle: the honey needs to remain in contact with teeth and gum tissue for a meaningful period rather than being immediately swallowed. The 2004 Otago study used a chewing format; the 2023 Zagreb trial applied honey subgingivally under clinical supervision; the Al-Kubaisi mouthwash study diluted honey for rinsing.
The grades used in oral health research correspond broadly to UMF 15+ and above, which in the MGO rating system is approximately MGO 514+. This reflects the minimum activity level at which consistent antibacterial effects have been observed. Our mānuka honey range covers MGO 250+ through to MGO 1050+. For oral health applications aligned with the research literature, MGO 514+ and above is the relevant starting point. Our MGO 1050+ (UMF 23+) is our highest-activity offering, independently tested by Hill Laboratories under IANZ accreditation.
Mānuka honey is a complement to, not a replacement for, regular brushing with fluoride toothpaste, flossing, and professional dental cleaning. The research supports it as an adjunct intervention with a plausible and increasingly well-documented mechanism. It does not replace mechanical plaque removal or professional periodontal treatment.
When to see your dentist
Mānuka honey is a bioactive food, not a dental treatment. The following symptoms require professional dental assessment:
- Toothache or persistent dental pain
- Visible cavity, broken, or chipped tooth
- Bleeding gums that persist despite improved oral hygiene
- Loose teeth or noticeable gum recession
- Swelling in the gum, jaw, or face
- Persistent bad breath that does not improve with good oral hygiene
The New Zealand Dental Association (nzda.org.nz) maintains a directory of registered practitioners. Regular check-ups remain the most evidence-based approach to long-term oral health. For more on the science of MGO and how it differs from other honey activity markers, see our guide: What Is MGO in Mānuka Honey? A Scientist's Complete Guide.
Frequently asked questions
Can mānuka honey reverse tooth decay?
No. Once enamel has demineralised to the point of forming a cavity, it cannot be reversed by honey or any other food product. Mānuka honey may support a healthier oral bacterial environment and, in combination with good oral hygiene, may help reduce the conditions driving decay. Existing cavities require professional dental treatment.
Which MGO grade is relevant for oral health?
Clinical research has generally used honey at UMF 15+ or higher, which corresponds to approximately MGO 514+ in the rating system used for export-certified New Zealand mānuka honey. If you are considering mānuka honey specifically for oral health use aligned with the published research, MGO 514+ and above is the appropriate starting point. Consult your dentist before changing your oral health routine.
Does mānuka honey cause cavities because of its sugar content?
Regular honey, used in quantity, contributes to bacterial acid production because its sugars are fermentable. High-MGO mānuka honey is different because its antibacterial activity counteracts the sugar-feeding effect. The 2004 University of Otago clinical study observed no increase in caries activity in the mānuka honey group over 21 days. Purposeful oral application is not the same as consuming large amounts. Context and quantity matter.
How do I use mānuka honey for my gums?
The clinical studies used different methods: a chewable honey preparation held in the mouth (Otago, 2004), honey applied subgingivally by a dental professional (Zagreb, 2023), and a diluted mouthwash formulation (Al-Kubaisi, 2023). There is no single standardised home protocol. The consistent principle is allowing the honey to remain in contact with gum tissue rather than swallowing immediately. Use this as a complement to brushing and flossing, not a replacement for either.
Does mānuka honey kill Streptococcus mutans?
In vitro and clinical evidence both support that high-MGO mānuka honey inhibits S. mutans growth and reduces salivary bacterial load. University of Otago in vitro research confirmed antibacterial activity against S. mutans specifically, and a 2023 randomised clinical trial demonstrated statistically significant reductions in salivary S. mutans levels after 21 days of mānuka honey mouthwash use [4]. Whether this translates to a meaningful long-term reduction in cavity incidence has not yet been established in large-scale trials.
Clinical Advisor, Tōtika Health Limited. Registered Nurse with expertise in health claim accuracy, consumer health content, and evidence-based framing of therapeutic evidence.
References
- English HKP, Pack ARC, Molan PC. The effects of manuka honey on plaque and gingivitis: a pilot study. J Int Acad Periodontol. 2004 Apr;6(2):63–7. PMID: 15125017
- Safii SH, Tompkins GR, Duncan WJ. Periodontal Application of Manuka Honey: Antimicrobial and Demineralising Effects In Vitro. Int J Dent. 2017;2017:9874535. doi: 10.1155/2017/9874535. PMC5368358.
- Vukovic A, et al. Therapeutic Manuka Honey as an Adjunct to Non-Surgical Periodontal Therapy: A 12-Month Follow-Up, Split-Mouth Pilot Study. Materials (Basel). 2023 Feb 1;16(3):1248. doi: 10.3390/ma16031248. PMC9921343.
- Al-Kubaisi MW, Al-Ghurabi BH. Evaluation of Manuka Honey Effects on Dental Plaque and Bacterial Load (Clinical Study). J Med Chem Sci. 2023;6(2):365–375. doi: 10.26655/JMCHEMSCI.2023.2.17
- Jungbauer G, Lechner R, Stähli A, Sculean A, Eick S. In-Vitro Effect of Manuka Honey / Propolis Toothpastes on Bacteria and Biofilm Associated with Caries and Gingivitis. Oral Health Prev Dent. 2025. doi: 10.3290/j.ohpd.c_1910. PMC11966148.






